19 April 2024
Best poster award - SiliconPV 2024
Congratulations to Olatz Arriaga Arruti for winning the Best Poster Award at the SiliconPV 2024 Conference! Explore her research on solar cell degradation mechanisms and mitigation solutions.
CSEM is recognized as an innovation vector in providing technology platforms that enable significant scientific and creative breakthroughs in today's world. Our products, solutions, and equality policies have been honored in Switzerland and around the world.

19 April 2024
Congratulations to Olatz Arriaga Arruti for winning the Best Poster Award at the SiliconPV 2024 Conference! Explore her research on solar cell degradation mechanisms and mitigation solutions.

27 November 2023
Congratulations to Laurie-Lou Senaud for her groundbreaking work and dedication to improving solar cell efficiencies, which resulted in her being awarded the Asea Brown Boveri Ltd. Award in 2023!

11 December 2022
Launched in March 2007, the Durabilis award is given to student projects that incorporate sustainability considerations. In 2022, it was awarded to Alexis Barrou for his project "Life Cycle Analysis & Sustainable Technological Roadmap for Photovoltaic Panels".

4 December 2022
MATIS, a Neuchâtel-based startup focusing on developing high-tech multispectral imaging systems, has received a financial support from the Foundation for Technological Innovation .

1 November 2022
Laurie-Lou Senaud, who won the EPFL's Best 8% Thesis Distinction award, conducted a scientific study focusing on the characterization and mitigation of electrical losses in silicon heterojunction (SHJ) solar cells.

30 March 2022
Arnaud Walter was rewarded with the SiliconPV 2022 Award for his presentation entitled "Perovskite/Silicon Tandems: First Realization with Rear Textured p-Type High-Temperature Passivating Contacts".
29 March 2022
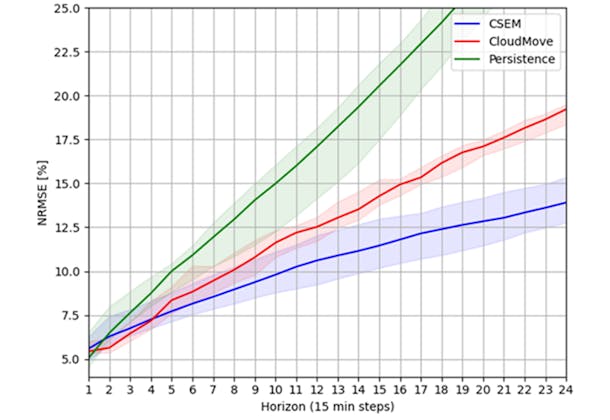
"Best Practice-Oriented Poster" goes to CSEM and Meteotest, for their work understanding the performance of data-driven and weather-based PV forecasting techniques.

11 March 2022
Congratulations to Ewelina Obrzud, winner of the prestigious European Astronomical Society's MERAC Prize for her amazing work on new instrumental technologies!

8 February 2022
A remarkable invention: an ultra-low-power transceiver, icyTRX, that can be integrated into Bluetooth chips, enabling greater miniaturization and a longer battery life for portable devices.
We meet the requirements and standards set by the Environmental management ISO 14001:2015, Management systems ISO 9001:2015, and Quality Management Systems for Medical Devices ISO 13485:2016. We are certified under these families, guaranteeing the quality of the products we produce for our clients.

SQS: ISO 9001 & 14001 - Quality management / Environmental management

IQNet: ISO 9001 & 14001 - Quality management / Environmental management

Bureau Veritas: ISO 13485 - Quality management / Medical devices